gCNG® — a Renewable Fuel from Biogas
Renewable fuels such as FirmGreen’s gCNG® offer a clean and economical alternative to fossil based fuels, derived from reliable, sustainable local resources.
FirmGreen® uses its patented and proprietary systems to convert biogas into highly purified ‘biomethane’ that exceeds pipeline standards. Biomethane is typically defined as a natural gas fuel from renewable resources—such as compressed natural gas (CNG), produced from waste resources such as landfills, waste water treatment plants, and livestock operations. FirmGreen’s technology successfully produces a biomethane product that meets OEM engine manufacturer specs for purity and Btu content.
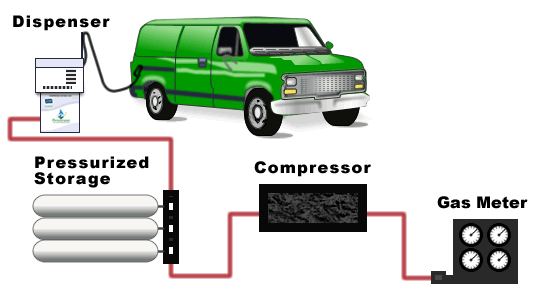
For a typical fleet fueling operation, FirmGreen technology is used to process landfill gas into ultra-pure biomethane, which can be compressed on-site for fueling CNG vehicles. Both time fill and fast fill dispensers can be installed to serve your fleet needs.
Why gCNG?
FirmGreen’s renewable compressed natural gas (gCNG®) fuel exceeds pipeline standards. is obtained by compressing purified biomethane and is stored and distributed in safe DOT approved containers.
Benefits of Landfill Gas-to-Biomethane:
- cleaner air benefits everyone
- gain control over annual fuel costs for your fleet
- gain a predictable domestic source for fuel
- turn your landfill methane into a revenue source
Benefits of FGI’s Compressed Natural Gas (gCNG®) Fleet Fueling Systems:
- CNG serves as an alternative to diesel or gasoline for motor vehicles. Due to its low hydrocarbon emissions, CNG is widely considered to be an environmentally viable alternative.
- Lower maintenance costs when vehicles run on CNG.
- Use of gCNG displaces fossil fuels and the cost of transporting petroleum based fuels to markets: give your business a competitive edge with cleaner fuels and limiting your exposure to petrol price volatility.
- Surveys have shown that natural gas vehicles (NGVs) are safer than gasoline-fueled vehicles.
- Vehicle life-span is prolonged due to cleaner combustion.
CNG or compressed natural gas is a domestically available, economical, clean burning, alternative fuel source for vehicles.
Rather than burn gasoline or diesel fuel, a consumer would fuel their vehicle with natural gas. In order to provide enough range, the gas is compressed and stored on the vehicle in pressurized tanks. Tanks that can hold up to 3,600 psig.
Sounds a little scary doesn’t it? Not when you compare the physical properties of Natural Gas and gasoline. The fact is that natural gas is a much safer fuel than gasoline. Natural gas readily disperses in air because of its simple molecular structure. (Ever wonder why the stove in your home doesn’t burn gasoline instead of natural gas?) Just take a look at the chart below and those on following tabs for some interesting facts:
Natural Gas – a simple hydrocarbon
A typical configuration consists of a pre-treatment skid, the biogas cleaning module, and optional polishing modules to condition the gas for specific applications. The system’s compact footprint makes it suitable for locations with limited land available to site a project.
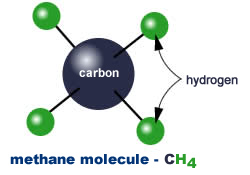
As you can see from the chart, natural gas has a ratio of four (4) hydrogen atoms to one (1) carbon atom. This makes CNG the lowest carbon fuel available today: with the exception of hydrogen itself, natural gas (NG) has the highest hydrogen content of any fuel.
Its simple molecule structure makes complete combustion easy and results in less tailpipe pollution than petroleum fuels. Natural Gas Vehicles (NGVs) produce approx. 93 – 95% less emissions than similar petroleum fueled vehicles. (source: EIA)

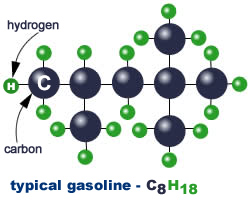 The second chart at left shows the molecular structure typical of gasoline – you can see that it’s much more complex than the simple structure of methane (natural gas) shown above at left . You’ll notice that there are eight (8) carbon atoms and eighteen (18) hydrogen atoms in each molecule. Because of the complexity of the molecule, gasoline tends not to completely combust in an engine.
The second chart at left shows the molecular structure typical of gasoline – you can see that it’s much more complex than the simple structure of methane (natural gas) shown above at left . You’ll notice that there are eight (8) carbon atoms and eighteen (18) hydrogen atoms in each molecule. Because of the complexity of the molecule, gasoline tends not to completely combust in an engine.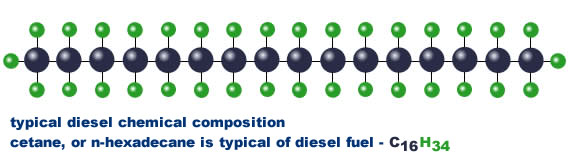 Along with carbon monoxide pollution resulting from incomplete combustion, other carbon atoms may remain stuck together with each other and with some of the hydrogen atoms also; these unburned hydrocarbon molecules come out of the tailpipe. The unburned hydrocarbons then react with nitrogen oxides (another pollutant from combustion) in the presence of sunlight to form ozone, which is a lung irritant.
Along with carbon monoxide pollution resulting from incomplete combustion, other carbon atoms may remain stuck together with each other and with some of the hydrogen atoms also; these unburned hydrocarbon molecules come out of the tailpipe. The unburned hydrocarbons then react with nitrogen oxides (another pollutant from combustion) in the presence of sunlight to form ozone, which is a lung irritant.